Study is first to compare Nexrutine to exercise for prostate cancer
Contact: Rosanne Fohn, (210) 567-3026, fohn@uthscsa.edu
SAN ANTONIO (Jan. 27, 2020) – Researchers from UT Health San Antonio are the first to show that Nexrutine, a natural supplement made from the bark of cork trees, is as effective as exercise in preventing the progression of aggressive prostate cancer.
Darpan Patel, Ph.D., associate professor of research in the School of Nursing, led the study, published Dec. 19 in the journal PLOS ONE. “Nexrutine is an herbal extract from the bark of the cork tree Phellodendron amurense. It is widely used to treat inflammation, gastroenteritis, abdominal pain and diarrhea,” Dr. Patel said.
In the study, supported by pilot funds from the Mays Cancer Center, home to UT Health San Antonio MD Anderson Cancer Center, 45 mice were divided into three groups. One group received food pellets supplemented with Nexrutine. The second group received free access to a running wheel. The third was the control group, which received no treatment.
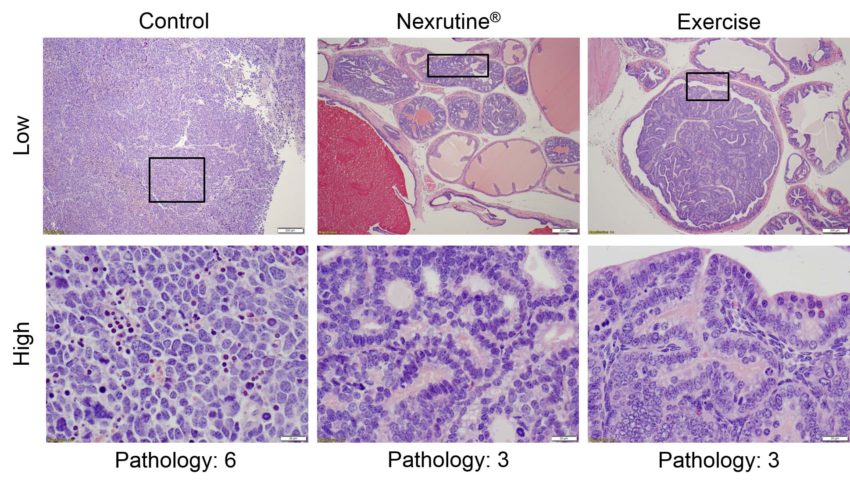
The overall results showed that the group receiving the cork tree extract had 62% less tumor growth, while the exercise group had 60% less tumor growth, compared to the control group. “It appears that both Nexrutine and exercise work equally well but use different pathways,” Dr. Patel said.
The research team came to this conclusion by evaluating the tumors and levels of various inflammatory markers in the blood after the 20-week study. They learned that mice in both treatment groups had a lower level of Interleukin 1 alpha (IL-1a), a type of protein that regulates inflammation and the expression of genes involved in the growth and survival of cancer cells, and the formation of abnormal blood vessels that support tumor growth. However, they were surprised to learn that the two treatment groups had some differences, indicating different pathways may be involved.
Pratap Kumar, Ph.D., professor in the UT Health San Antonio Department of Molecular Medicine, was a key investigator on the study. Dr. Kumar has carried out extensive work with Nexrutine in various cancers including a clinical study in prostate cancer patients.
Dr. Patel said that more research will be needed to better understand the separate mechanisms and how they work. “Perhaps a combination treatment could be developed in the future that would attack the cancer on several fronts,” he said. “Nexrutine also may be a good alternative for people who find it difficult to exercise,” Dr. Kumar said.
Both Drs. Patel and Kumar are members of the Population Sciences and Prevention program of Mays Cancer Center, a National Cancer Institute-designated cancer center. Dr Kumar is also co-leader of this program.“The results of these research studies by Mays Cancer Center investigators is deeply exciting. If confirmed in subsequent human clinical trials, the discovery of this new way to control aggressive prostate cancer is an important addition to our arsenal against cancer,” said Mays Cancer Center Director Ruben Mesa, M.D., FACP.
According to the American Cancer Society, prostate cancer is the second most common type of cancer in men behind skin cancer and is the second-leading cause of cancer deaths in men after lung cancer.
# # #
For current news from the UT Health Science Center San Antonio, now called UT Health San Antonio®, please visit our online newsroom, like us on Facebook or follow us on Twitter.
# # #
The University of Texas Health Science Center at San Antonio, now called UT Health San Antonio®, is one of the country’s leading health sciences universities. With missions of teaching, research, healing and community engagement, its schools of medicine, nursing, dentistry, health professions and graduate biomedical sciences have produced 36,500 alumni who are leading change, advancing their fields and renewing hope for patients and their families throughout South Texas and the world. To learn about the many ways “We make lives better®,” visit www.uthscsa.edu.