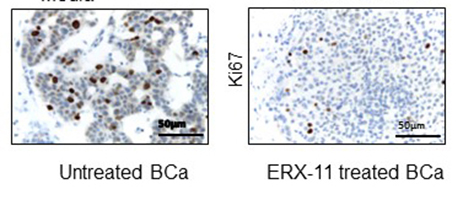
SAN ANTONIO (Aug. 9, 2017) ― Researchers at UT Health San Antonio and two partner institutions are developing a new, first-in-class agent that has stopped the growth of estrogen receptor-positive breast cancer in its tracks. The new agent is a molecule called ERX-11 that has blocked the growth of recurring breast cancer tumors.
Ratna Vadlamudi, Ph.D., is principal investigator of a study describing the new findings, published Aug. 8 in the journal eLIFE. He is professor of obstetrics and gynecology in the Joe R. & Teresa Lozano Long School of Medicine and is a member of the Cancer Development & Progression Program (CDP) at the UT Health Cancer Center.
“Most breast cancers in women require estrogen or progesterone – the two female hormones — to grow. To treat hormone-dependent cancers, you must either block the hormone or block the receptor that receives the hormone to prevent the development of the cancer. However, in many patients, tumors become resistant to the current therapy and tumors recur. Our team at UT Health discovered a new molecule that blocks the estrogen receptor signaling that occurs in resistant tumors and tested a drug based on the molecule in preclinical studies in the lab,” Dr. Vadlamudi said.
“Developing this drug is important because it targets a unique site on the estrogen receptor and blocks its interactions with critical proteins that contribute to breast cancer progression. This drug will be useful in treating breast cancers that are sensitive to current therapies and that are resistant to therapy,” he said.
Researchers at UT Dallas led by Jung-Mo Ahn, Ph.D., were involved in synthesizing the drug that blocked the interaction, and scientists at UT Southwestern led by Ganesh Raj, M.D., Ph.D., are involved in mechanistic studies and in studies involving primary human breast tumors.
“Implanting mice with the human breast cancer cells usually causes the mice to develop the breast tumors, but when we gave the mice ERX-11 as an oral medication, the cancer growth was dramatically reduced,” Dr. Vadlamudi said. The studies in mice showed no toxic effects, indicating the drug based on the ERX-11 molecule would be a good candidate for human trials.
“The next steps are to synthesize the drug under the FDA’s drug development guidelines and conduct formal toxicity studies to ensure the drug can be safely tolerated,” Dr. Vadlamudi said. “Additional work needs to be done before we can move forward with Phase I clinical trials. We expect human trials to begin soon.”
UT Health San Antonio holds a joint patent on the estrogen receptor-positive breast cancer drug with UT Southwestern and UT Dallas, with the UT Southwestern Office for Technology Development managing development of the drug.
Other researchers from UT Health San Antonio include Rajeshwar Tekmal, Ph.D., professor, and Gangadhara Reddy Sareddy, Ph.D., assistant professor, both from the UT Cancer Center CDP and Department of Obstetrics & Gynecology; and Suryavathi Viswanadhapalli, Ph.D., a post-doctoral fellow. Monica Mann, Ph.D., and Samaya Rajeshwari Krishnan, Ph.D., former graduate students, also worked on the study.
These studies were funded with a $1.9 million grant from the Cancer Prevention Research Institute of Texas to UT Health in November 2014 . Drs. Raj and Ahn were co-principal investigators of this grant.
# # #
For current news from the UT Health Science Center San Antonio, now called UT Health San Antonio™, please visit our online newsroom, like us on Facebook or follow us on Twitter.
# # #
The University of Texas Health Science Center at San Antonio, with missions of teaching, research and healing, is one of the country’s leading health sciences universities and is now called/doing business as UT Health San Antonio™. UT Health San Antonio’s schools of medicine, nursing, dentistry, health professions and graduate biomedical sciences have produced more than 33,000 alumni who are advancing their fields throughout the world. With four campuses in San Antonio and Laredo, UT Health San Antonio has a FY 2017 revenue operating budget of $806.6 million and is the primary driver of its community’s $37 billion biomedical and health care industry. For more information on the many ways “We make lives better®,” visit www.uthscsa.edu.