Novel AI algorithm precisely quantifies perivascular spaces that signal risk of stroke.
An artificial intelligence (AI) tool developed at The University of Texas Health Science Center at San Antonio accurately counts brain lesions on MRI scans within seconds. Once it is adapted to the clinic, the AI tool should help neuroradiologists to evaluate patients’ brain diseases at earlier stages.
“Certain kinds of brain lesions are tremendously difficult to quantify without AI,” said researcher Mohamad Habes, PhD, of the health science center’s Glenn Biggs Institute for Alzheimer’s and Neurodegenerative Diseases. Habes is assistant professor of radiology and director of the Biggs Institute neuroimaging core.

In a study published April 24 in JAMA Network Open, Habes and colleagues from eight institutions demonstrated the AI tool’s utility in identifying and counting enlarged perivascular spaces (ePVS’s). These spaces, filled with cerebrospinal fluid, surround arteries and veins and are a marker of cerebral small-vessel disease that can lead to stroke and dementia. The study was a follow-up analysis of 1,026 individuals who participated in the Multi-Ethnic Study of Atherosclerosis (MESA).
“We have developed an innovative deep-learning tool to precisely quantify every single enlarged perivascular space in the brain and provide us with a map of the patient’s small-vessel disease,” Habes said.
Taking a painstaking task and making it possible
Before this tool, ePVS’s were ignored because of the difficulty of counting them on MRI scans.
“On average a middle-aged person might have maybe 500 or 600 of these small spaces on an MRI,” Habes said. “Think about a neuroradiologist who is going to sit down and count all of them. That’s not really going to happen. He or she would spend one or two hours per scan, or even more, and that amount of laborious effort is not feasible in the workflow of a busy clinic.”
The team described the automated deep-learning method for ePVS detection in the journal Neuroimage: Reports on March 7. “We have trained an algorithm with expert knowledge to be able to quantify these lesions on its own,” Habes said. “This tool recognizes them, tells us their exact locations, counts them and tells us their volumes. It tells us a ton of information about them, far more than what a human can do.”
In the JAMA Network Open report, Habes and coauthors studied enlarged perivascular spaces throughout the brain.
“Before, people were not able to do whole-brain ePVS quantification,” Habes said. “We can now do it with our advanced deep learning tools. In our study, we realized that enlarged perivascular spaces in two regions, the basal ganglia and the thalamus, are the most significant lesions because they showed association with stroke and small-vessel disease markers.”
The basal ganglia is a deep-brain region important for neurodegenerative disorders and is related to movement and decision-making, Habes said. The thalamus, a region near the basal ganglia, is related to sensory functions such as taste and touch.
The researchers’ hope is that the AI tool for enumerating brain lesions will be further studied at the Alzheimer’s Disease Research Centers (ADRCs), which are U.S. National Institute on Aging-Designated Centers of Excellence. The South Texas ADRC, the only such center in Texas, is a collaboration of UT Health Science Center San Antonio’s Biggs Institute with The University of Texas Rio Grande Valley.
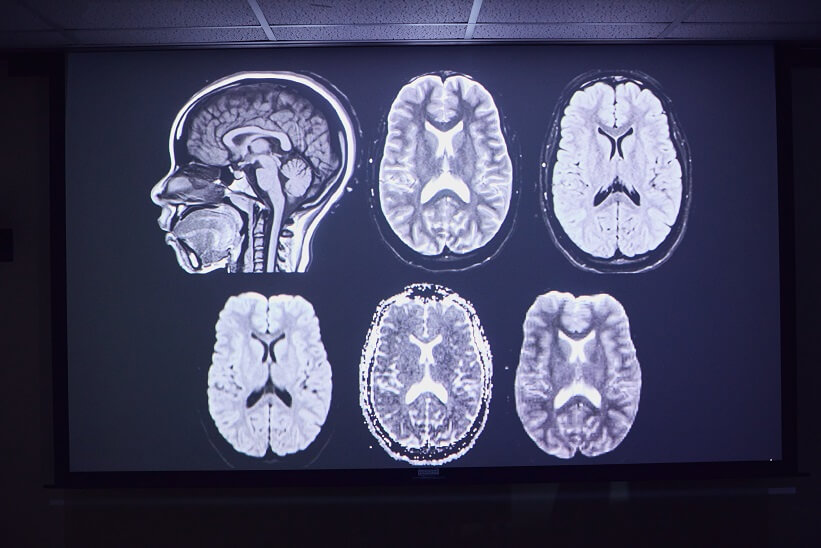 “This is a great breakthrough for our ADRC, which is focusing a lot on cerebrovascular disease and its contribution to dementia,” Habes said. “This is one of the unique themes of our ADRC, and we think our novel AI methodology can benefit large-scale studies conducted across the nation’s ADRCs.”
“This is a great breakthrough for our ADRC, which is focusing a lot on cerebrovascular disease and its contribution to dementia,” Habes said. “This is one of the unique themes of our ADRC, and we think our novel AI methodology can benefit large-scale studies conducted across the nation’s ADRCs.”
The AI tool leverages the power of UT Health Science Center San Antonio’s Genie supercomputer, Habes said.
Assessment of Risk Factors and Clinical Importance of Enlarged Perivascular Spaces by Whole-Brain Investigation in the Multi-Ethnic Study of Atherosclerosis
Sokratis Charisis, MD; Tanweer Rashid, PhD; Hangfan Liu, PhD; Jeffrey B. Ware, MD; Paul N. Jensen, PhD; Thomas R. Austin, PhD; Karl Li, MD, PhD; Elyas Fadaee, MD; Saima Hilal, MD, PhD; Christopher Chen, MD; Timothy M. Hughes, PhD; Jose Rafael Romero, MD; Jon B. Toledo, MD, PhD; Will T. Longstreth Jr, MD; Timothy J. Hohman, PhD; Ilya Nasrallah, MD, PhD; R. Nick Bryan, MD, PhD; Lenore J. Launer, PhD; Christos Davatzikos, PhD; Sudha Seshadri, MD; Susan R. Heckbert, MD, PhD; Mohamad Habes, PhD
First published: JAMA Network Open, April 24, 2023
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2803993